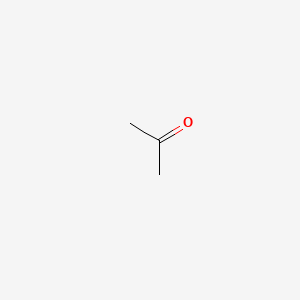
similar ffer. jum eride Which statements are true regarding these two compounds? Check all that apply. - brainly.com

SOLVED: Given the following aqueous solutions A: acetone, CH3COCH3 B: silver nitrate, AgNO3 C: ethylene glycol, HOCH2CH2OH D: sulfuric acid, H2SO4 E: ammonium bromide, NH4Br Which of these conduct electricity well?

Performance of a packed-bed anode bio-electrochemical reactor for power generation and for removal of gaseous acetone - ScienceDirect

Applied Sciences | Free Full-Text | Effect of Acetone Content on the Preparation Period and Curing/Pyrolysis Behavior of Liquid Polycarbosilane

Dissociative Adsorption of Acetone on Platinum Single-Crystal Electrodes | The Journal of Physical Chemistry C

SOLVED: Similar offer. Jump ride. Which statements are true regarding these two compounds? Check all that apply. Acetone is a covalent compound, while sodium chloride is an ionic compound. Ionic compounds conduct
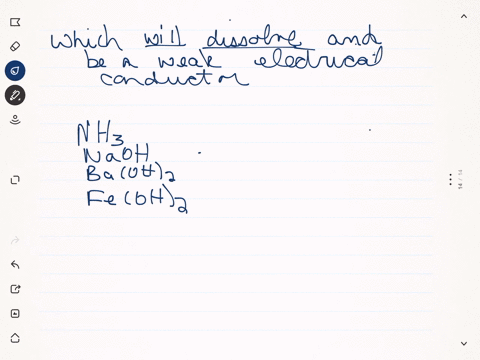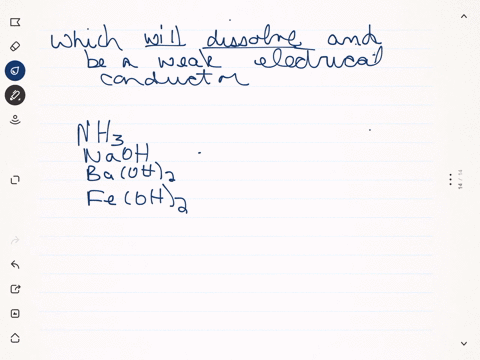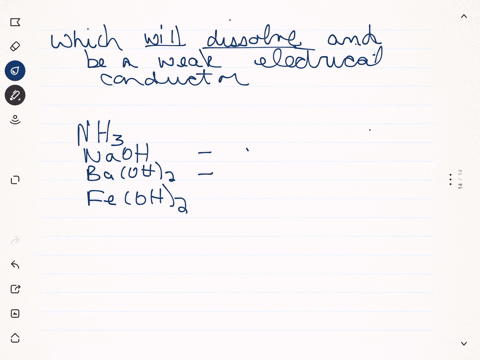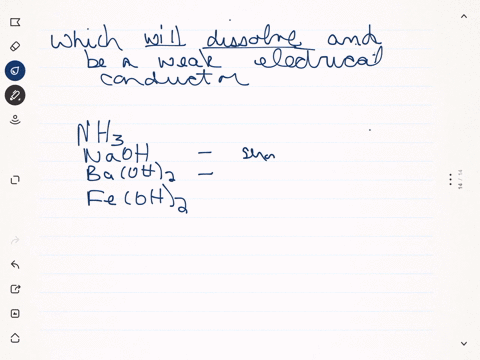
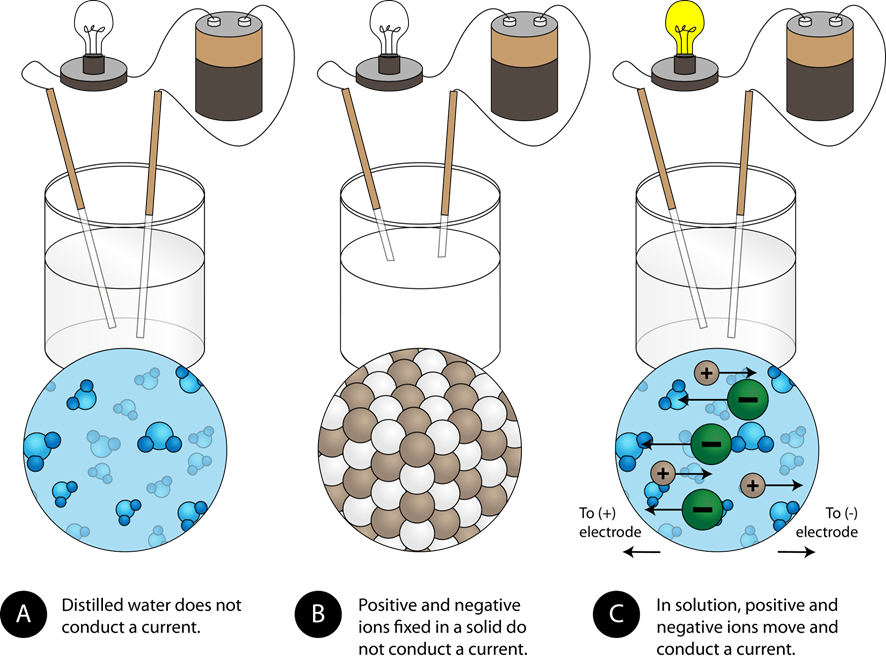
SOLVED:Predict whether each substance listed will conduct electricity, dissolve in water, and/ or conduct electricity once it has dissolved. Explain your thinking in each case. a. C3H6O(l) acetone b. Ti(s) titanium c.

Chapter 4 - General Chemistry - ▫ 6: multiply the empirical formula by this integer to determine - Studocu

similar ffer. jum eride Which statements are true regarding these two compounds? Check all that apply. - brainly.com
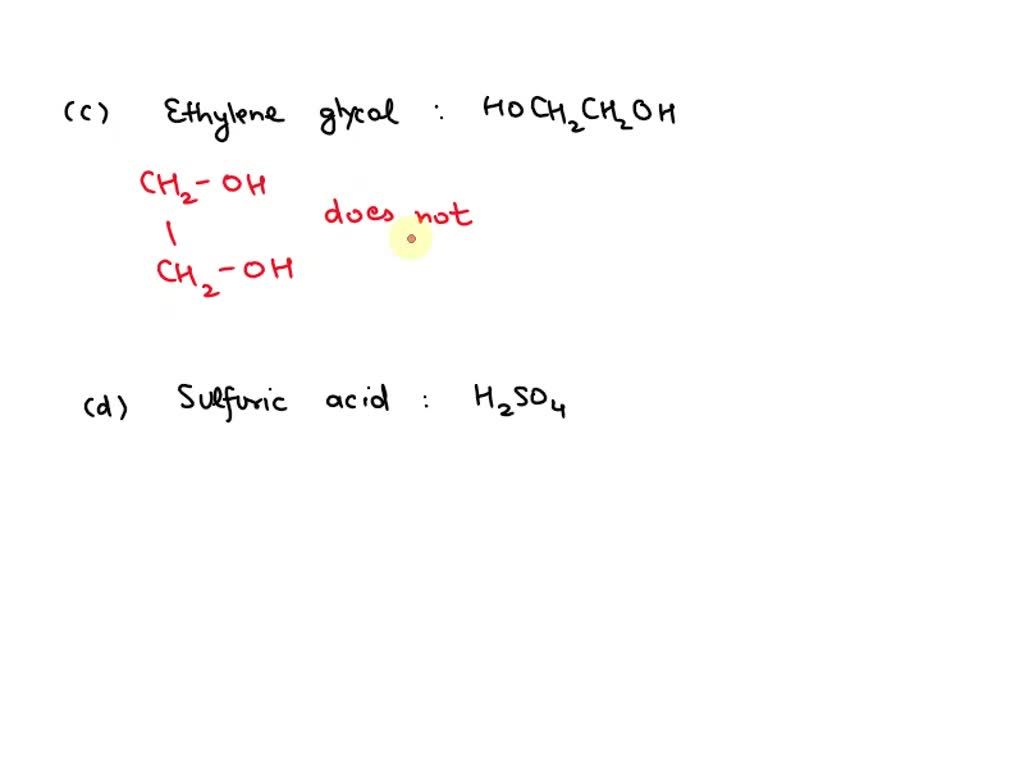
SOLVED: Given the following aqueous solutions A: acetone, CH3COCH3 B: silver nitrate, AgNO3 C: ethylene glycol, HOCH2CH2OH D: sulfuric acid, H2SO4 E: ammonium bromide, NH4Br Which of these conduct electricity well?
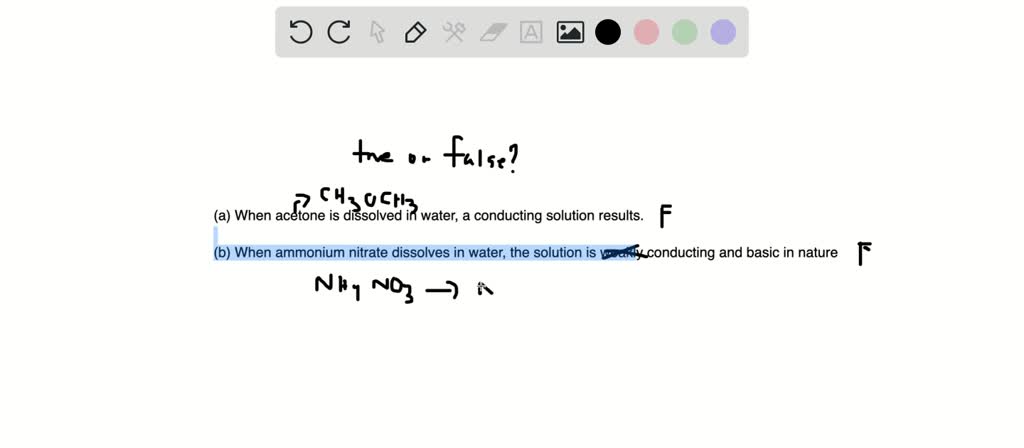
SOLVED:State whether each of the following statements is true or false. Justify your answer in each case. (a) When acetone, CH3 COCH3, is dissolved in water, a conducting solution results. (b) When

Dissociative Adsorption of Acetone on Platinum Single-Crystal Electrodes | The Journal of Physical Chemistry C