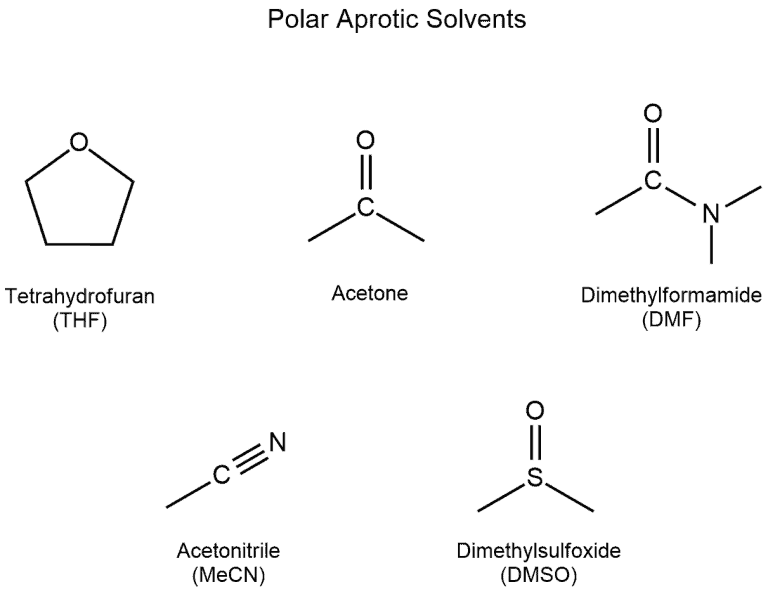
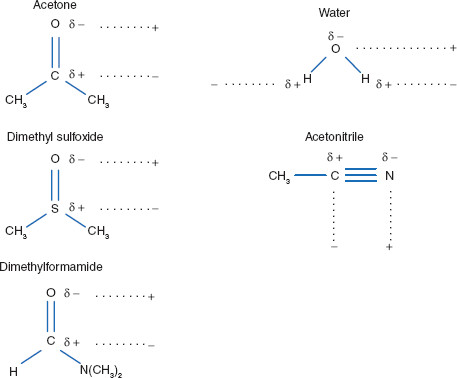
From: Acetone, Dimethsulfoxide, Dimethylformamide, water, HMPA, Ethanol, Acetic acid, DMA. The number of polar aprotic solvent in the following solvents is .

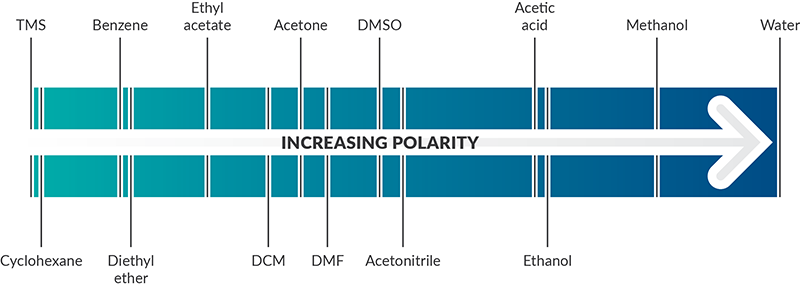
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com

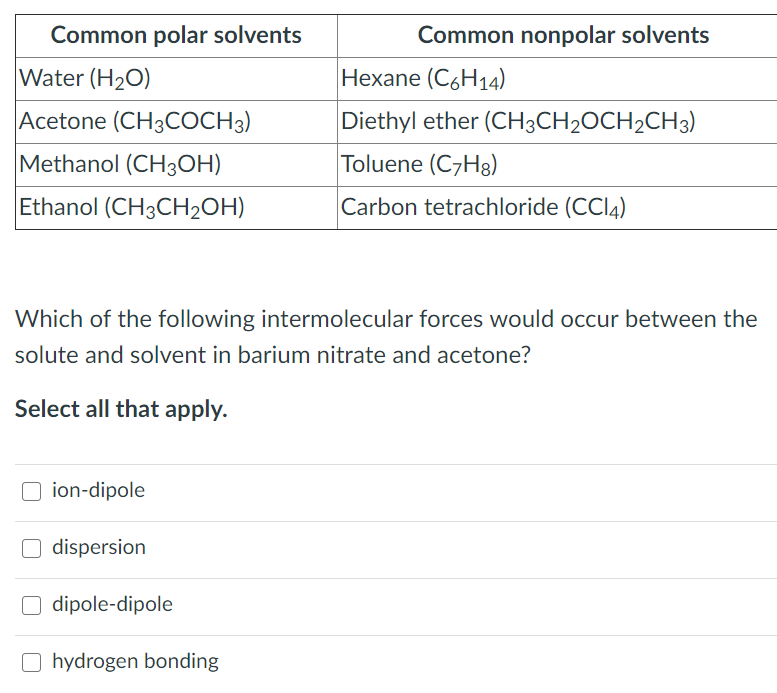
SOLVED: Although it is straightforward to predict the solubility of simple molecules, some molecules may be soluble in both polar solvents like water and nonpolar solvents such as hexane. One such example
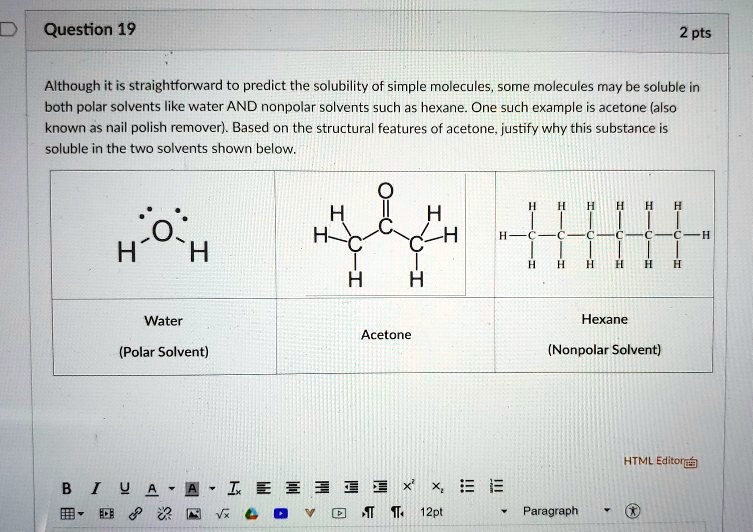
SOLVED: Although it is straightforward to predict the solubility of simple molecules, some molecules may be soluble in both polar solvents like water and nonpolar solvents such as hexane. One such example

Vector illustration of a metal liquid container can with acetone C3H6O. Illustration of aliphatic polar chemical solvent. On the packaging is the name and formula of the chemical substance.:: موقع تصميمي

A study of the effects of the polarity of the solvents acetone and cyclohexane on the luminescent properties of tryptophan - ScienceDirect
What makes acetone a really good solvent? What allows it to dissolve both polar and non-polar molecules? - Quora