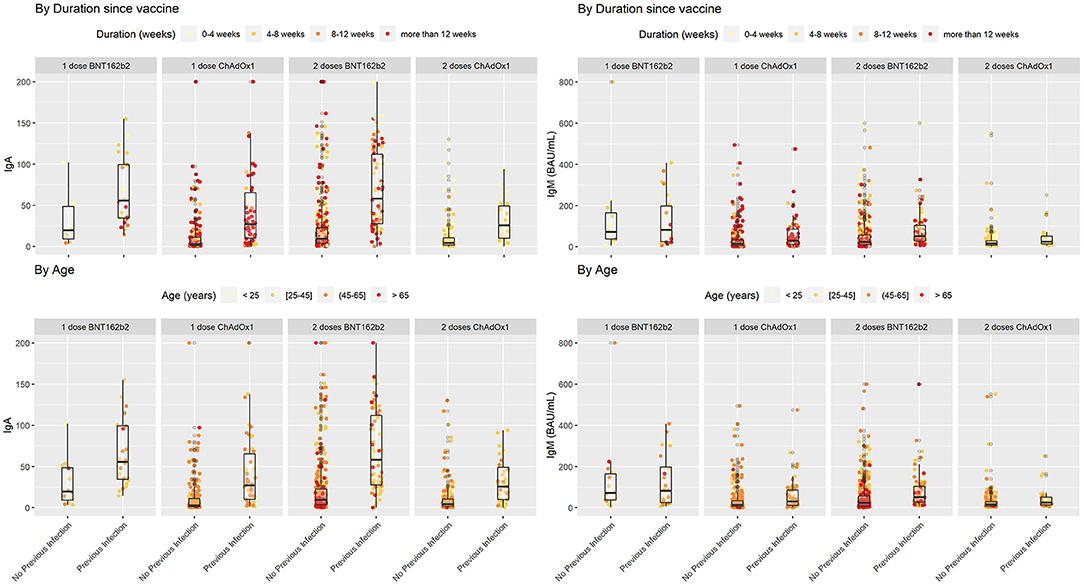
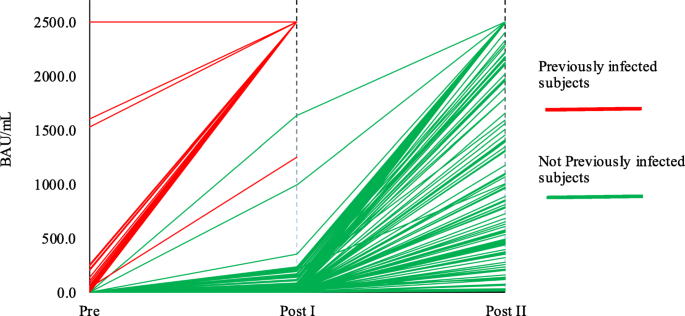
The comparability of Anti-Spike SARS-CoV-2 antibody tests is time-dependent: a prospective observational study | medRxiv
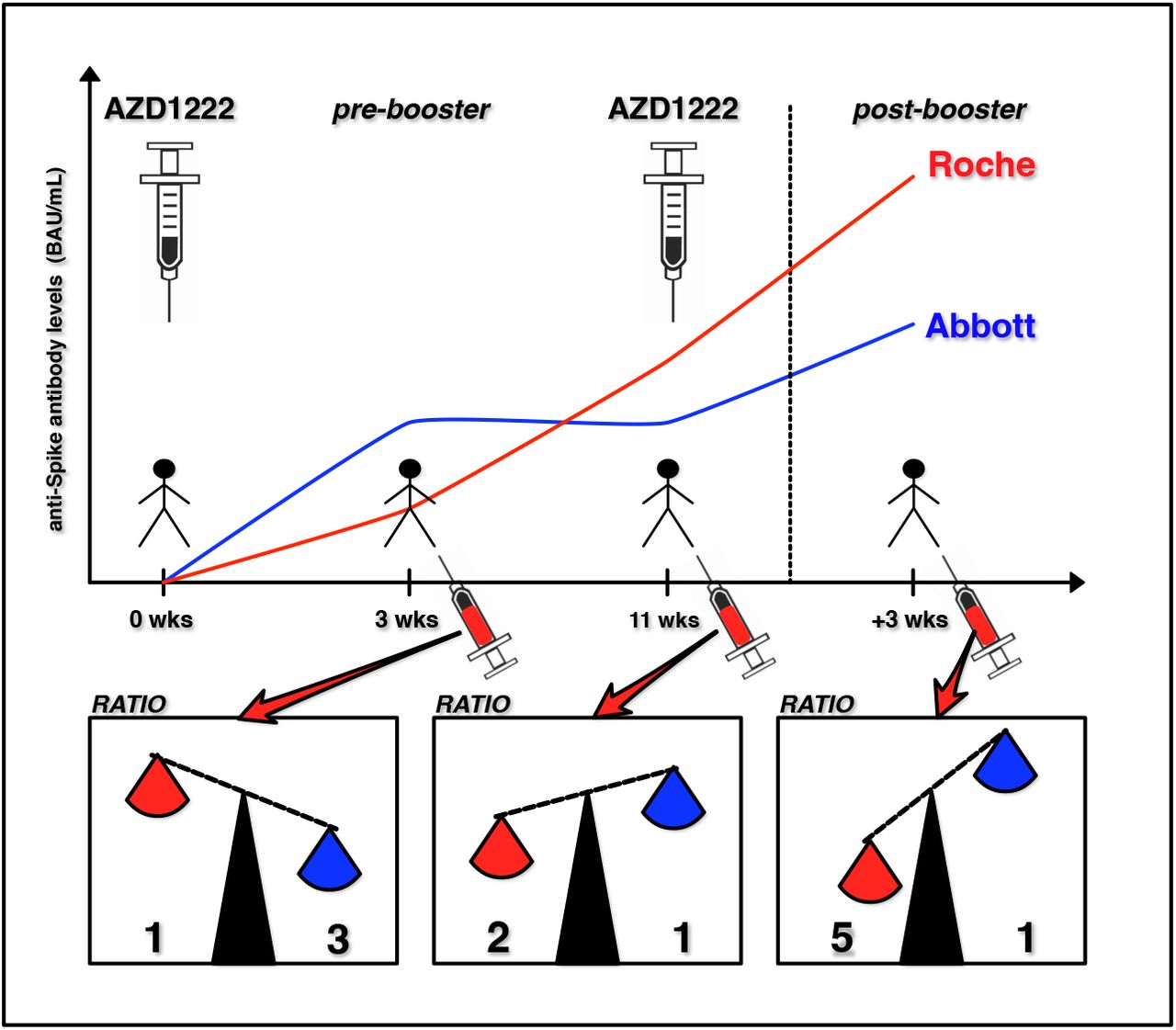
Vaccination strategy and anti - SARS-CoV-2 S titers in healthcare workers of the INT – IRCCS “Fondazione Pascale” Cancer Center (Naples, Italy) | Infectious Agents and Cancer | Full Text
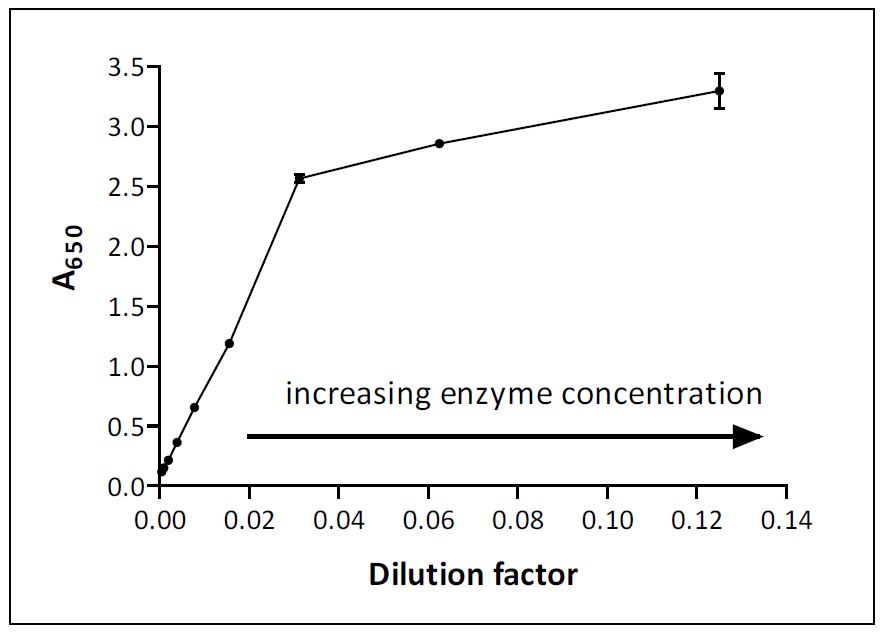
Guide to Enzyme Unit Definitions and Assay Design | Biomol Blog | Resources | Biomol GmbH - Life Science Shop

COVID-19 vaccine booster dose needed to achieve Omicron-specific neutralisation in nursing home residents - eBioMedicine

Evolution of SARS-CoV-2 spike antibodies (U/mL) in seronegative (blue)... | Download Scientific Diagram

Frontiers | IgG Anti-Spike Antibodies and Surrogate Neutralizing Antibody Levels Decline Faster 3 to 10 Months After BNT162b2 Vaccination Than After SARS-CoV-2 Infection in Healthcare Workers
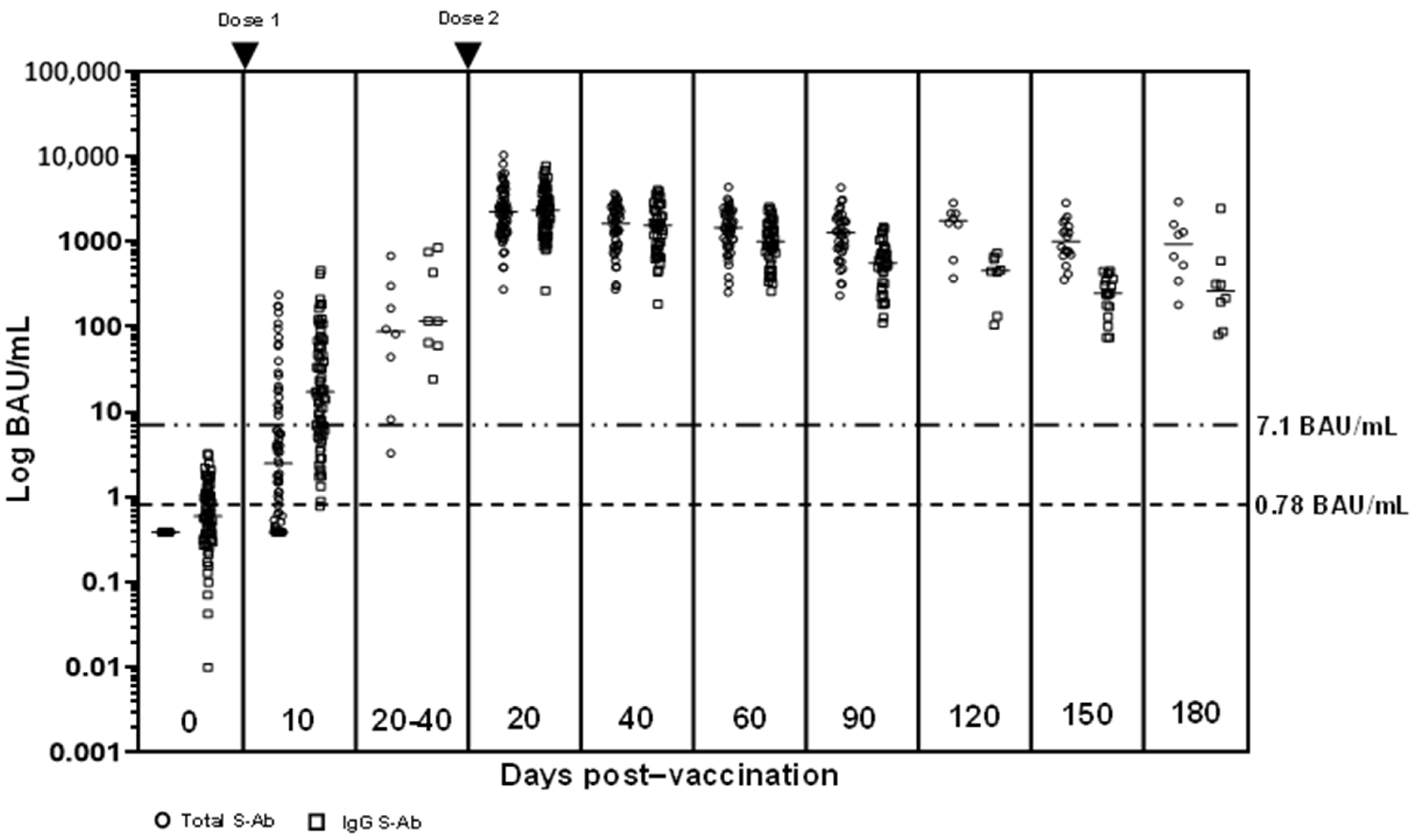
Vaccines | Free Full-Text | Robust SARS-CoV-2 Antibody Responses in Asian COVID-Naïve Subjects 180 Days after Two Doses of BNT162b2 mRNA COVID-19 Vaccine

Estimating immunity with mathematical models for SARS-CoV-2 after COVID-19 vaccination | npj Vaccines
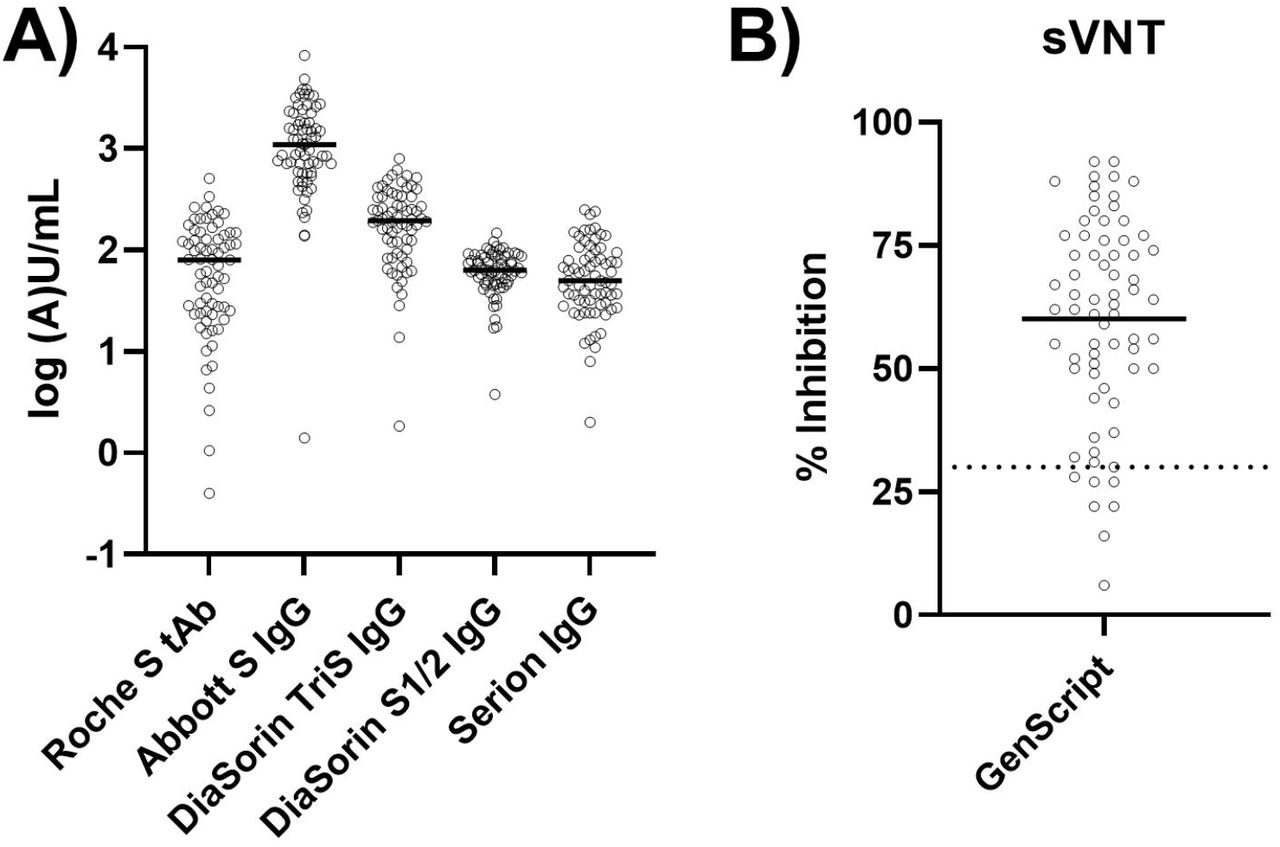
Anti-Spike protein assays to determine post-vaccination antibody levels: a head-to-head comparison of five quantitative assays | medRxiv

Evolution of antibody responses up to 13 months after SARS-CoV-2 infection and risk of reinfection - eBioMedicine
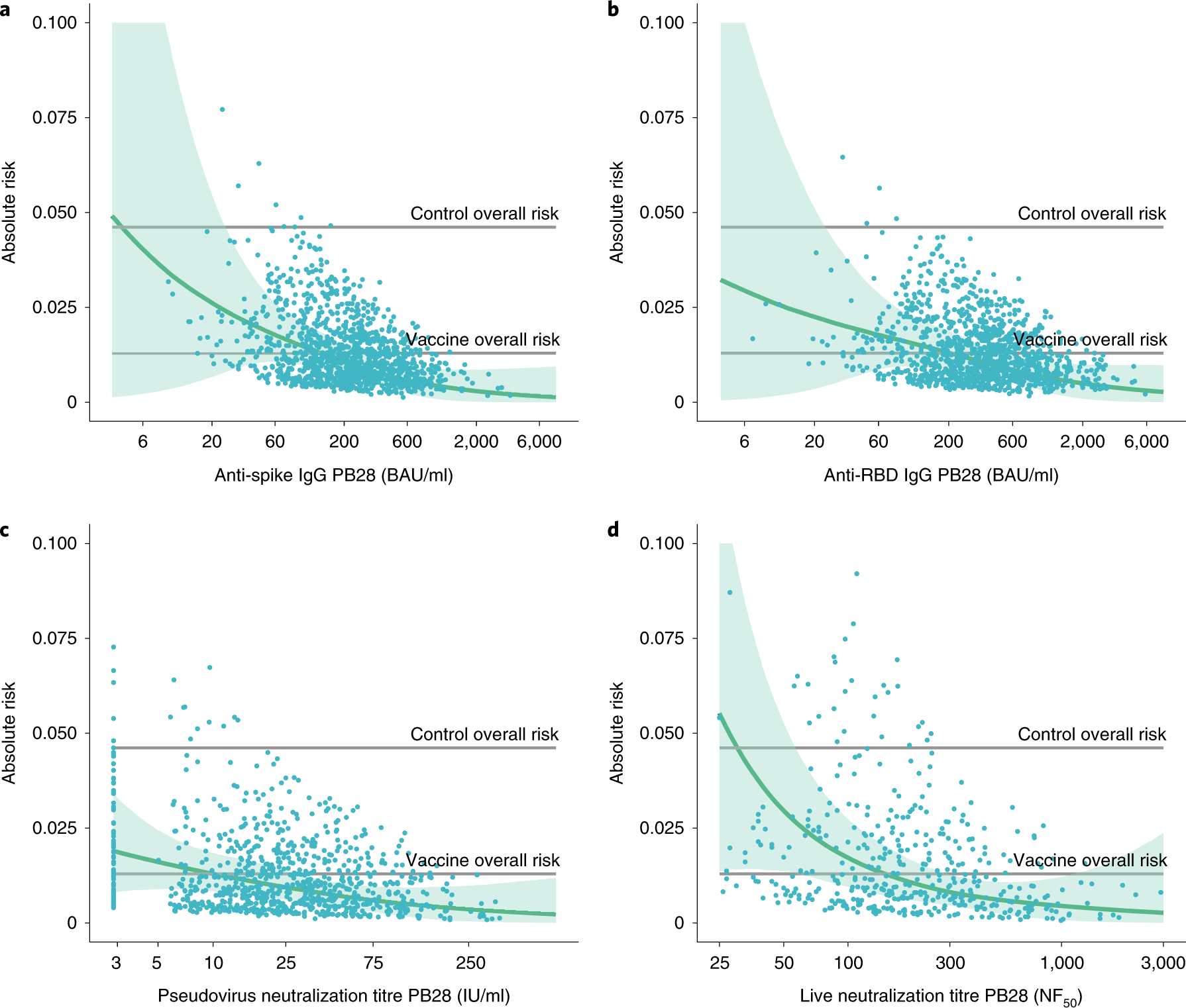
Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection | Nature Medicine
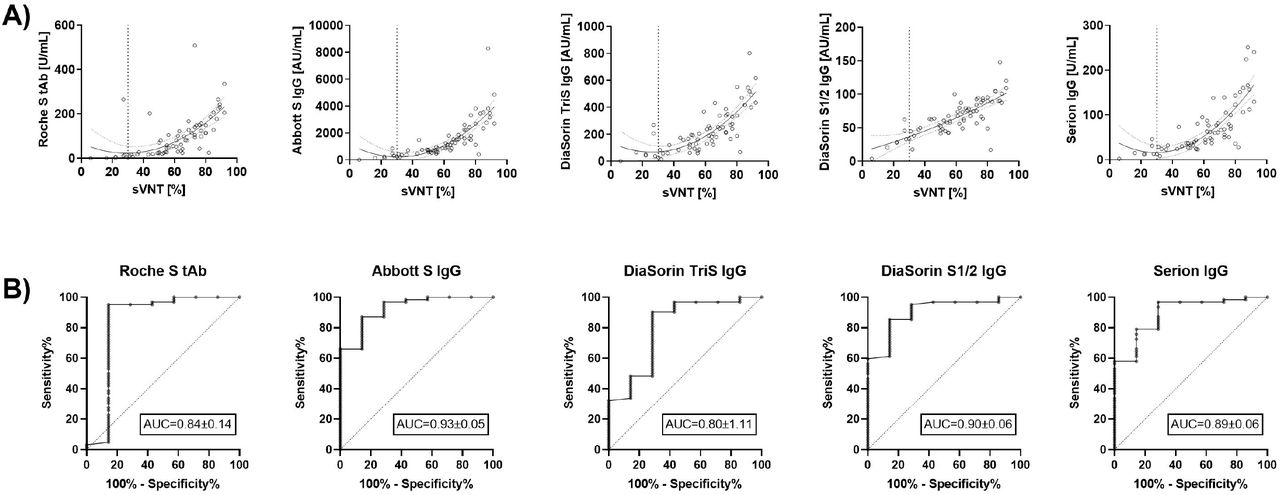
Anti-Spike protein assays to determine post-vaccination antibody levels: a head-to-head comparison of five quantitative assays | medRxiv

Frontiers | Clinical Utility of Elecsys Anti-SARS-CoV-2 S Assay in COVID-19 Vaccination: An Exploratory Analysis of the mRNA-1273 Phase 1 Trial

WHO International Standard for evaluation of the antibody response to COVID-19 vaccines: call for urgent action by the scientific community - The Lancet Microbe

Inhibition of microbial deconjugation of micellar bile acids protects against intestinal permeability and liver injury | Science Advances