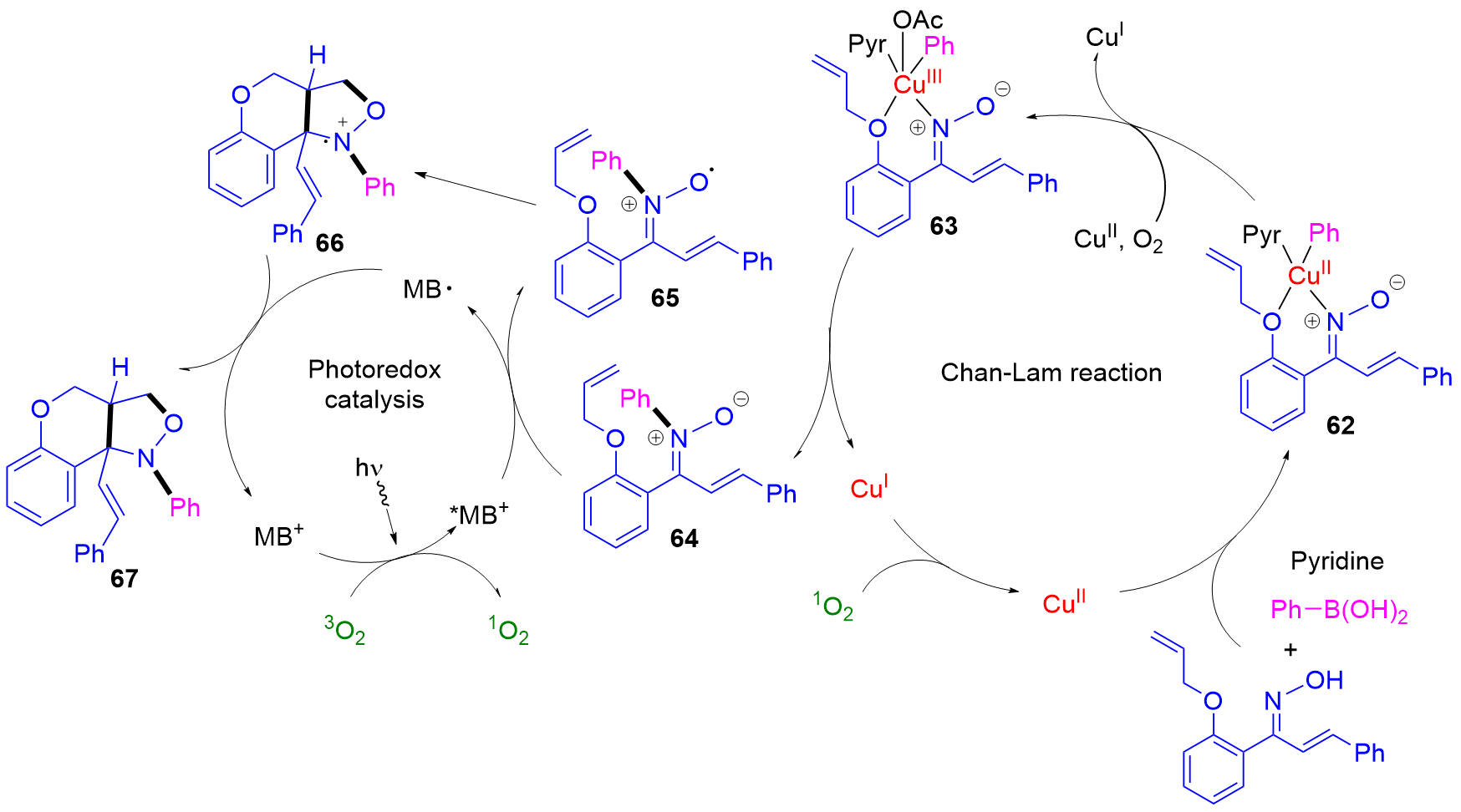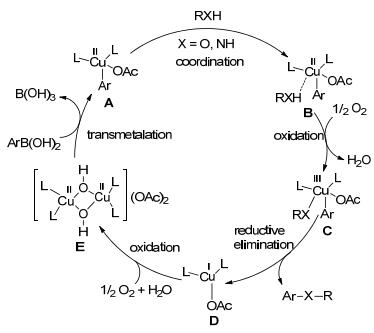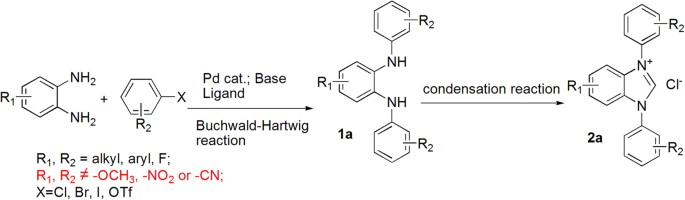
Application of Chan-Lam cross coupling for the synthesis of N-heterocyclic carbene precursors bearing strong electron donating or withdrawing groups | Scientific Reports

A room temperature one-pot Knoevenagel-Chan-Evans-Lam coupling reaction for synthesis of N-aryl-2-Iminocoumarins in bio-mass-derived green solvent 2-methylTHF - ScienceDirect

Visible‐Light‐Mediated Chan–Lam Coupling Reactions of Aryl Boronic Acids and Aniline Derivatives - Yoo - 2015 - Angewandte Chemie International Edition - Wiley Online Library

A Review on the Latest Progress of Chan‐Lam Coupling Reaction - Chen - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
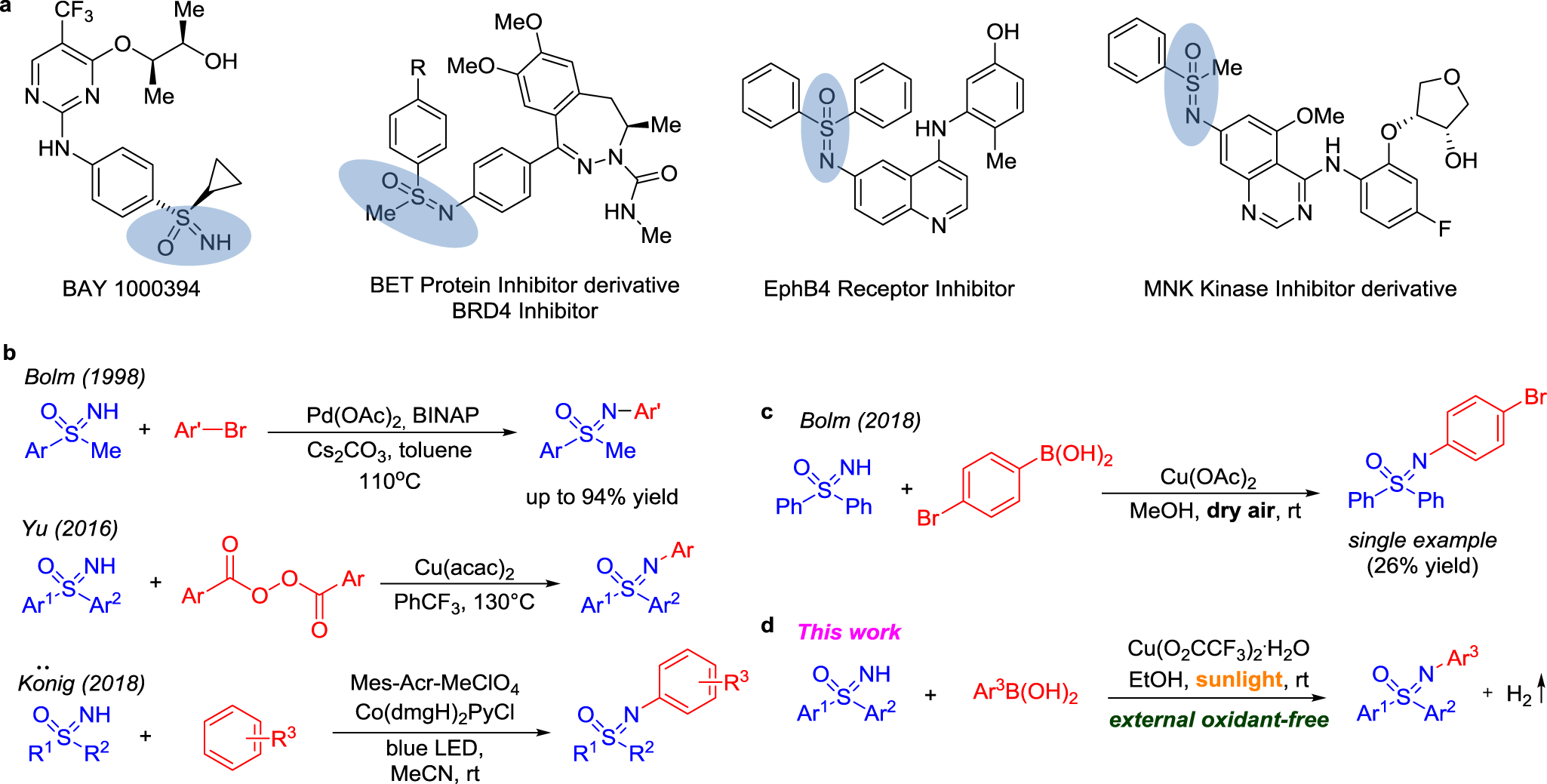
Autocatalytic photoredox Chan-Lam coupling of free diaryl sulfoximines with arylboronic acids | Nature Communications

A Review on the Latest Progress of Chan‐Lam Coupling Reaction - Chen - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
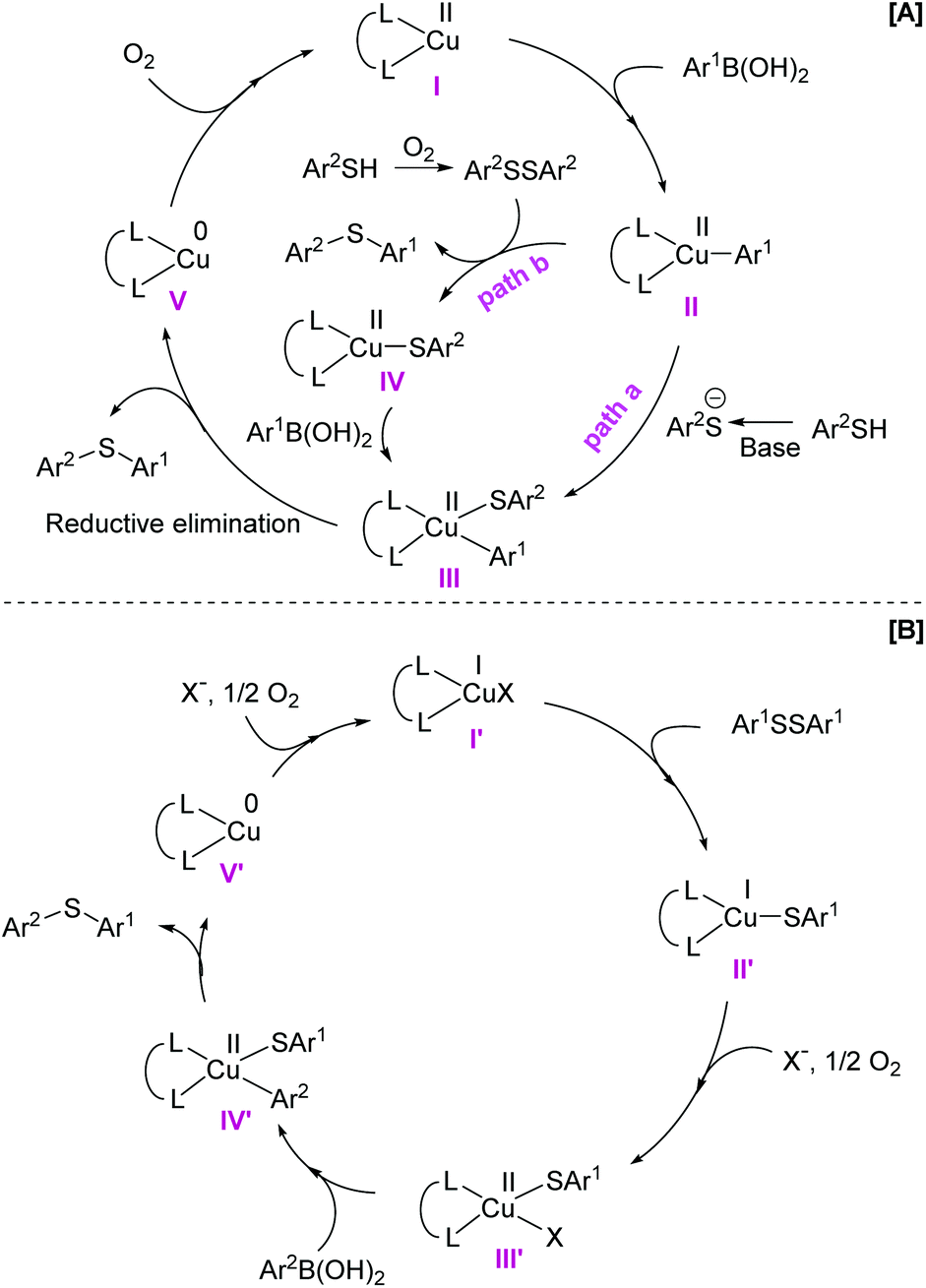
Advances in Cu and Ni-catalyzed Chan–Lam-type coupling: synthesis of diarylchalcogenides, Ar 2 –X (X = S, Se, Te) - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB02035D
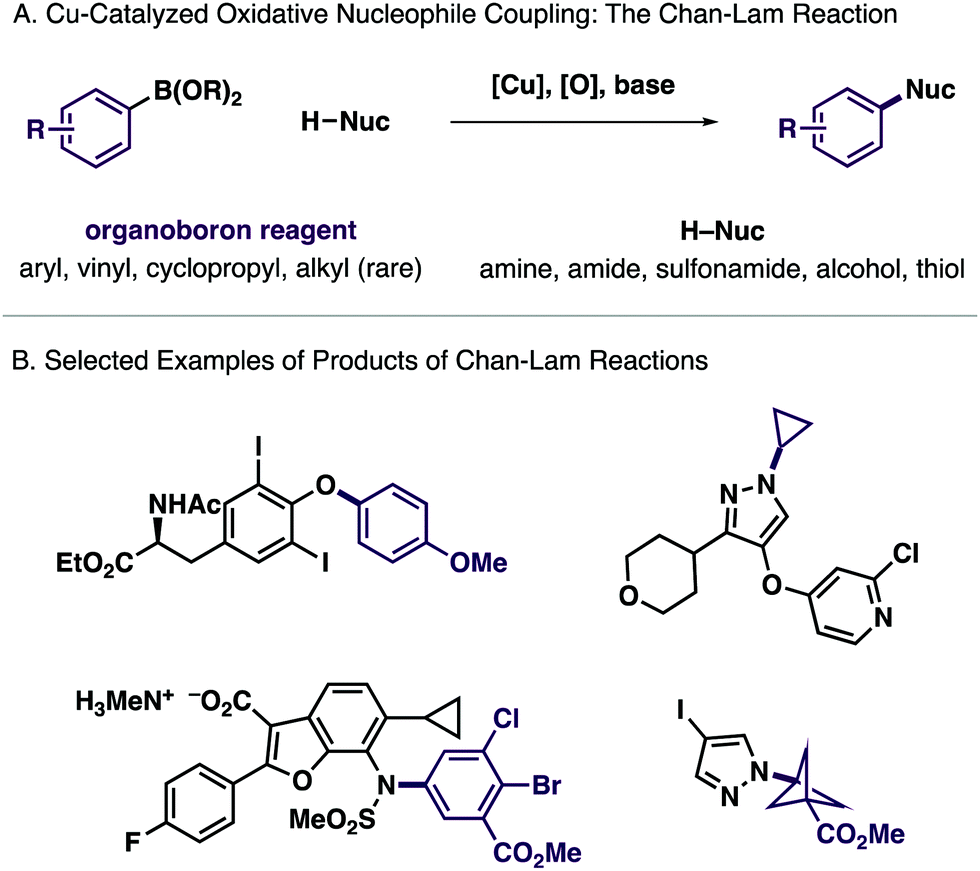
Oxidative cross-coupling processes inspired by the Chan–Lam reaction - Chemical Communications (RSC Publishing) DOI:10.1039/D1CC00213A

Chemoselective Chan–Lam Coupling Reactions between Benzimidazoline-2-thiones and Arylboronic Acids | The Journal of Organic Chemistry

Spectroscopic Studies of the Chan–Lam Amination: A Mechanism-Inspired Solution to Boronic Ester Reactivity | Journal of the American Chemical Society


![14] Chan-Evans-Lam Cross Coupling 1998 – ChemInfoGraphic 14] Chan-Evans-Lam Cross Coupling 1998 – ChemInfoGraphic](https://cheminfographic.files.wordpress.com/2017/08/14-chan_evans_lam_cross_coupling.jpg)