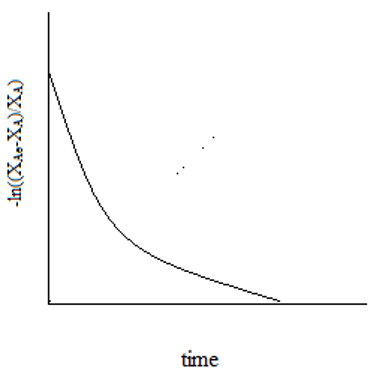
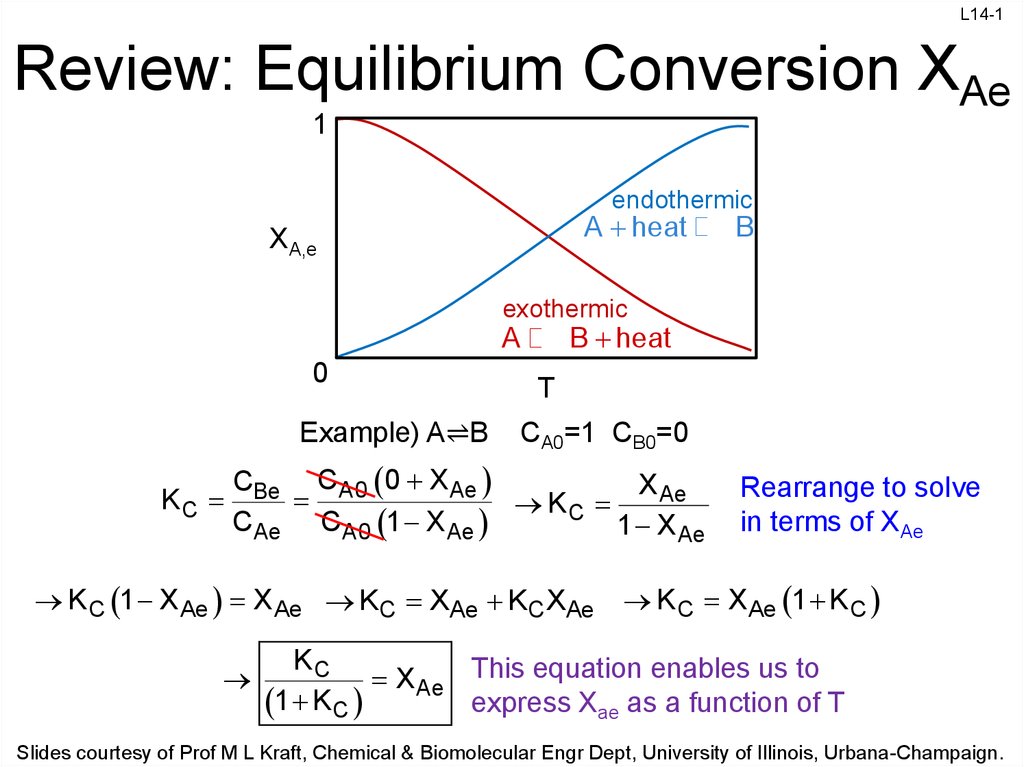
P8-8 The elementary gas phase reaction A B + C is carried out adiabatically in PFR packed with catalyst. Pure A enters the reactor at a volumetric flow. - ppt download

a) Equilibrium conversion of CO 2 and equilibrium selectivities of (b)... | Download Scientific Diagram

Methanol equilibrium conversion versus temperature in the MH + MeOH =... | Download Scientific Diagram

Equilibrium conversion when the pressure is 50 atm, Kp = 0.0016 by using Excel Goal Seek | TeacherOn

Equilibrium conversion vs equilibrium constant, at various reactants ratio | Download Scientific Diagram

A) Conversion B) Temperature C) Equilibrium constant and D) Reaction... | Download Scientific Diagram

4: Equilibrium conversion for the dehydrogenation of ethane, propane,... | Download Scientific Diagram

SOLVED: (a) Between 0°C and 100°C, determine the equilibrium conversion for the elementary aqueous reaction. Here, assume that ΔHxn is not a strong function of T. That is, you can use ΔH298k