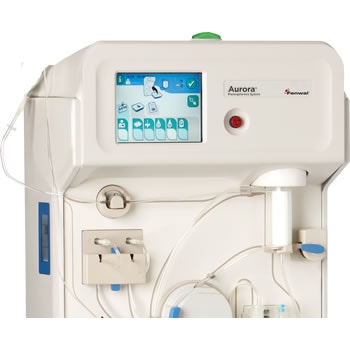
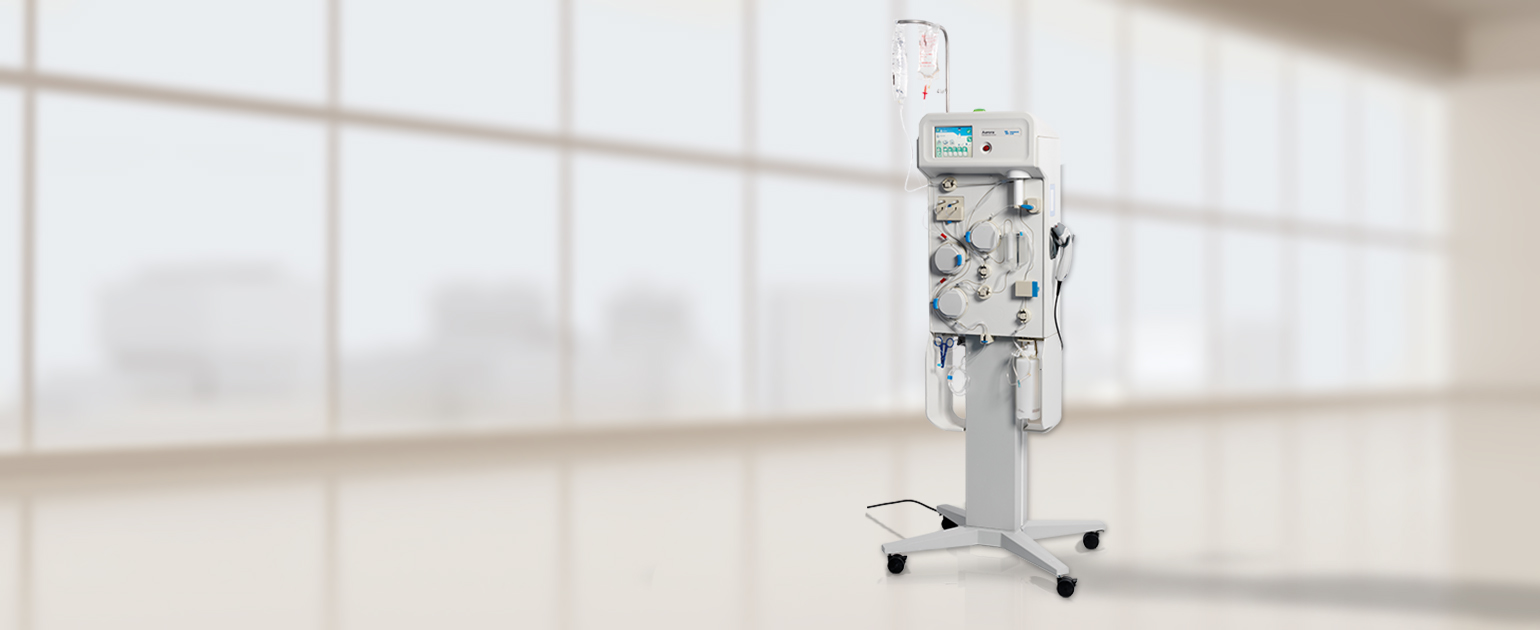
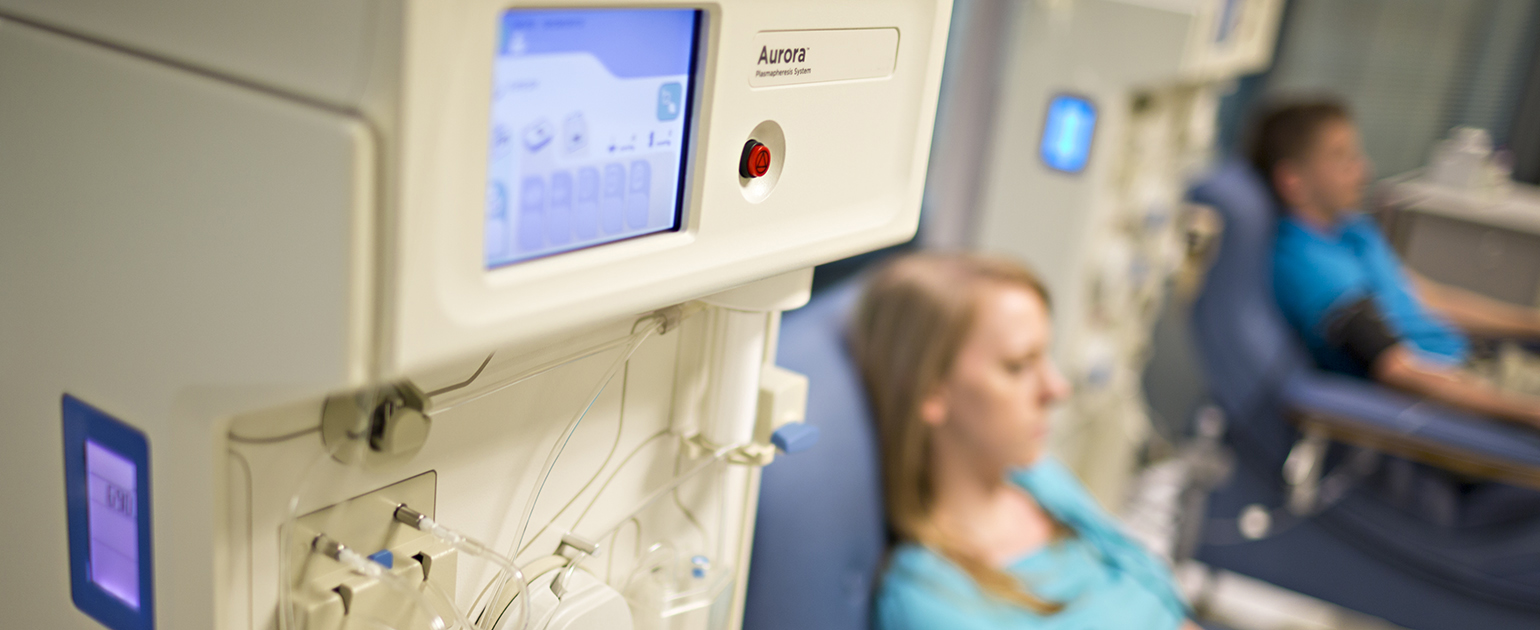
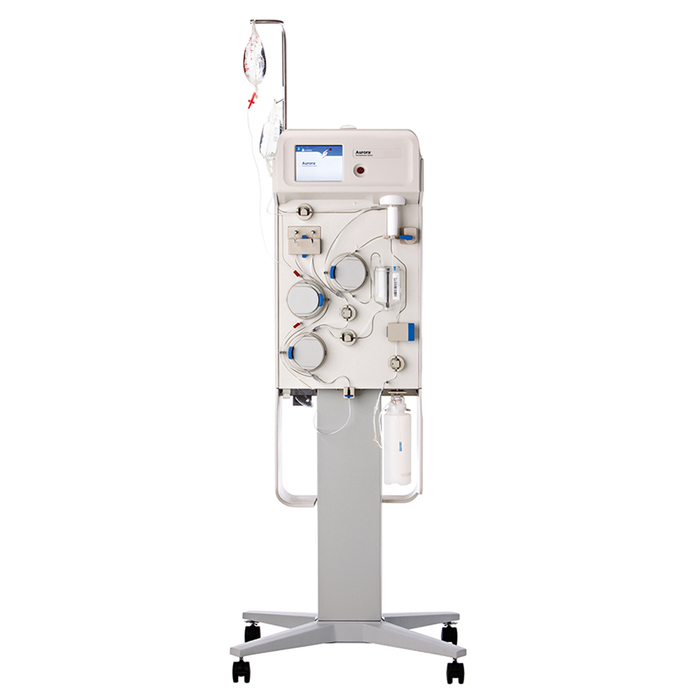
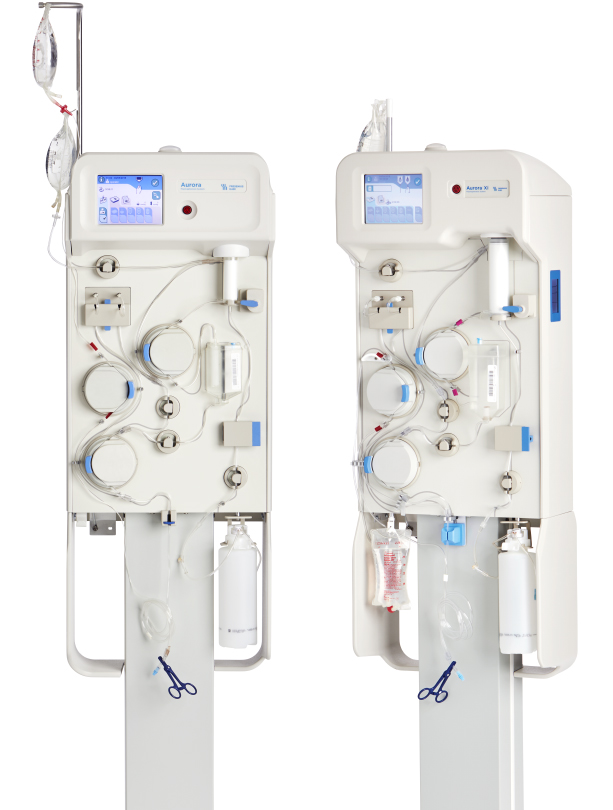
Fresenius Kabi Receives FDA 510(k) Clearance for the Aurora™ Xi Plasmapheresis System | Business Wire

aHUS Alliance - New Plasmapheresis Machine Hits Market Fresenius Kabi announced 7 Oct it has gained FDA 510(k) clearance for its Aurora Xi plasmapheresis system, designed to speed up filtration with improved