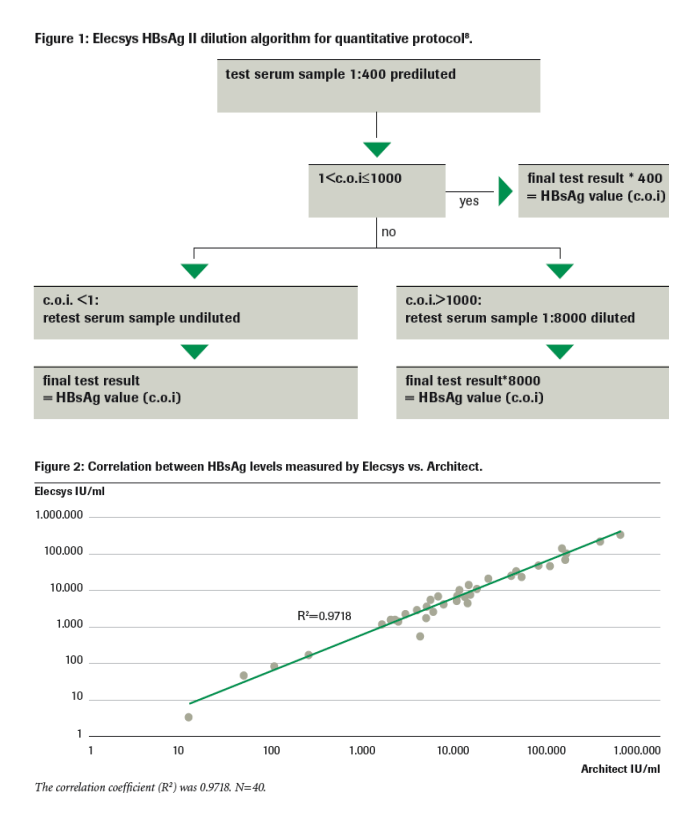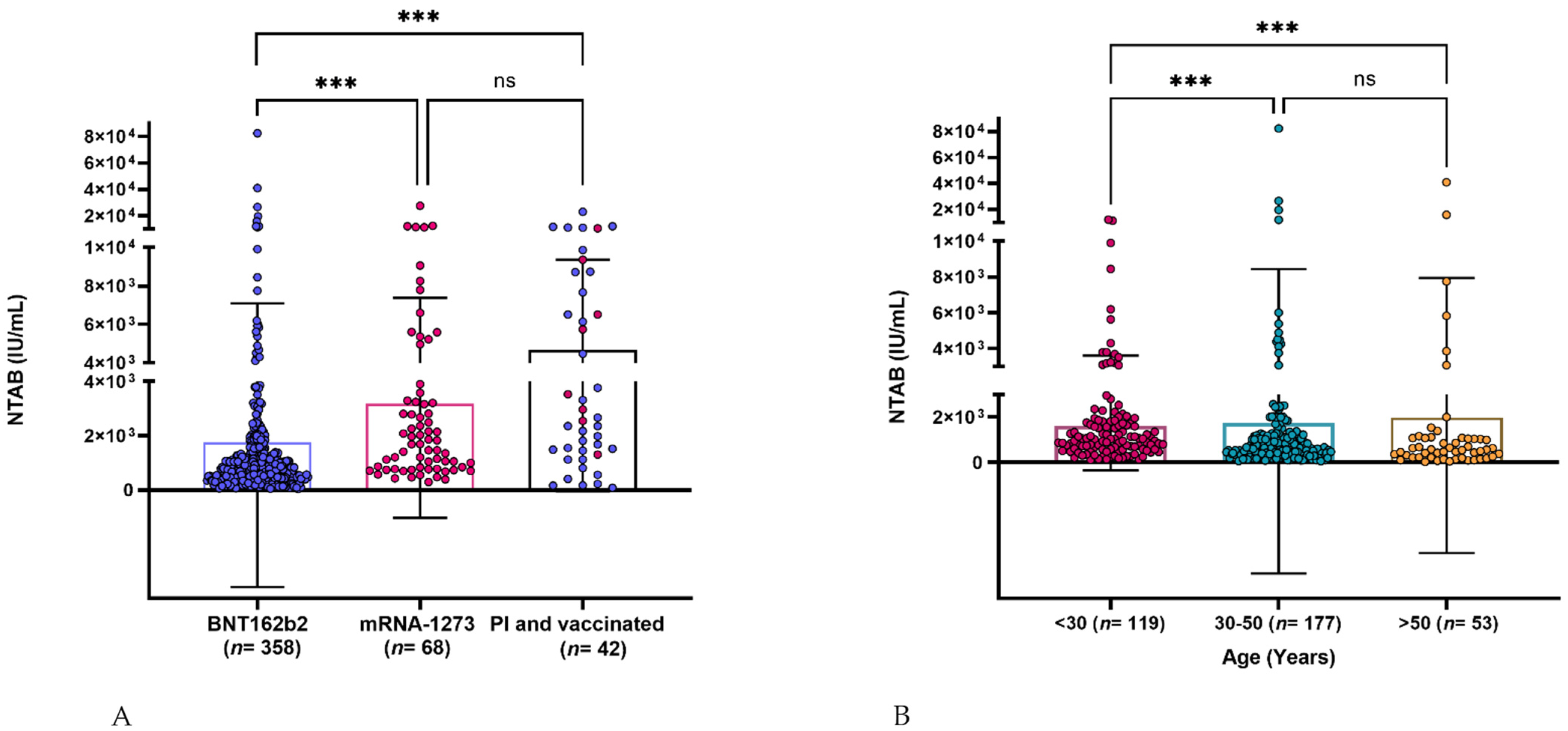
Vaccines | Free Full-Text | Assessment of the Neutralizing Antibody Response of BNT162b2 and mRNA-1273 SARS-CoV-2 Vaccines in Naïve and Previously Infected Individuals: A Comparative Study

Multicenter Evaluation of Whole-Blood Epstein-Barr Viral Load Standardization Using the WHO International Standard | Journal of Clinical Microbiology

Commutability of Cytomegalovirus WHO International Standard in Different Matrices | Journal of Clinical Microbiology
GitHub - Lelouchzhu/cPass-to-IU_Conversion: cPass reading (% inhibition) to International Units (IU)/ml of the WHO International Standard