Variation of effectiveness with mass flow rate of hot fluid (T hi =... | Download Scientific Diagram

SOLVED: Acetone has an experimentally determined latent heat of vaporization of 30.2 kJ/mol. Use different sources of latent heat and verify with the given experimental value, with the use of Perry's Chemical

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram
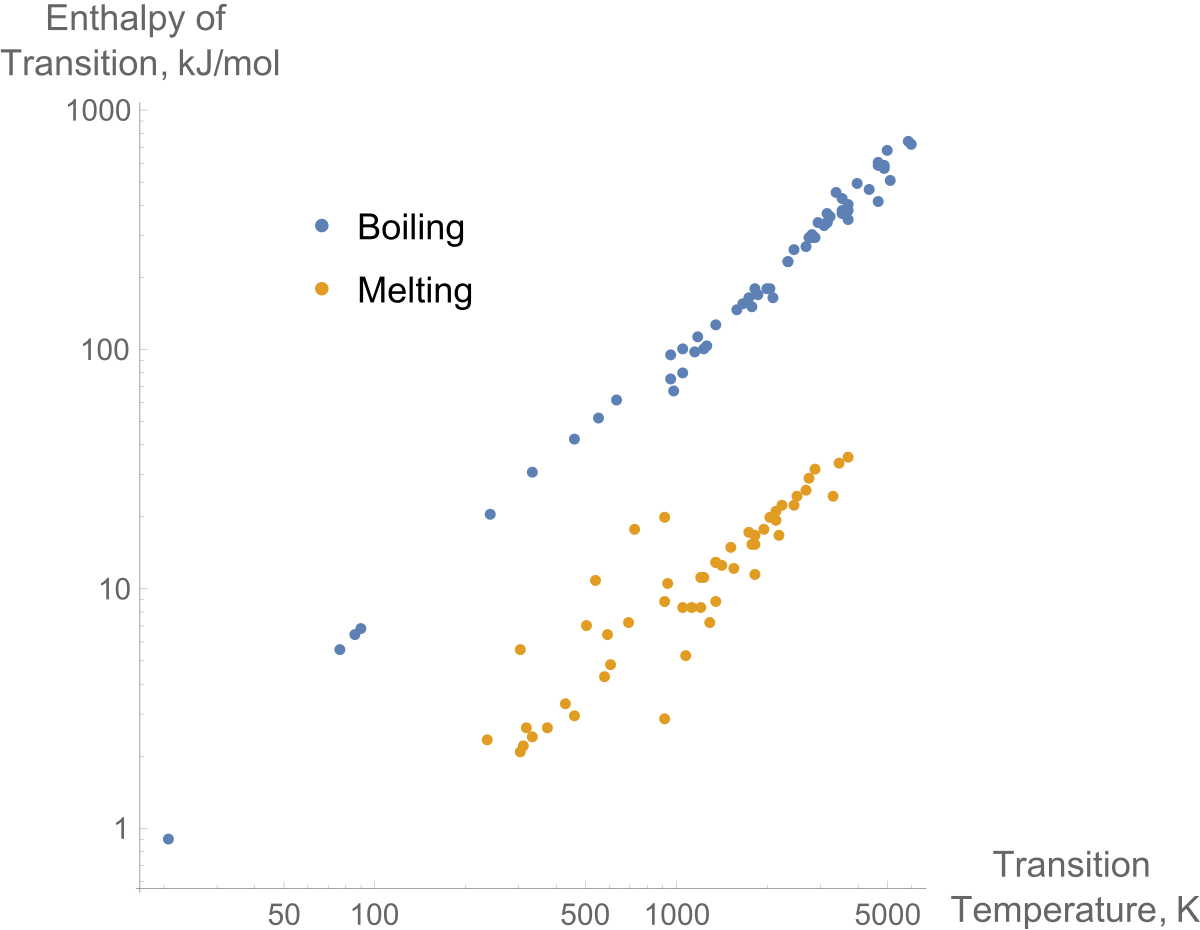
A Theoretical Analysis on Enthalpy of Vaporization: Temperature-Dependence and Singularity at the Critical State Abstract 1. Int
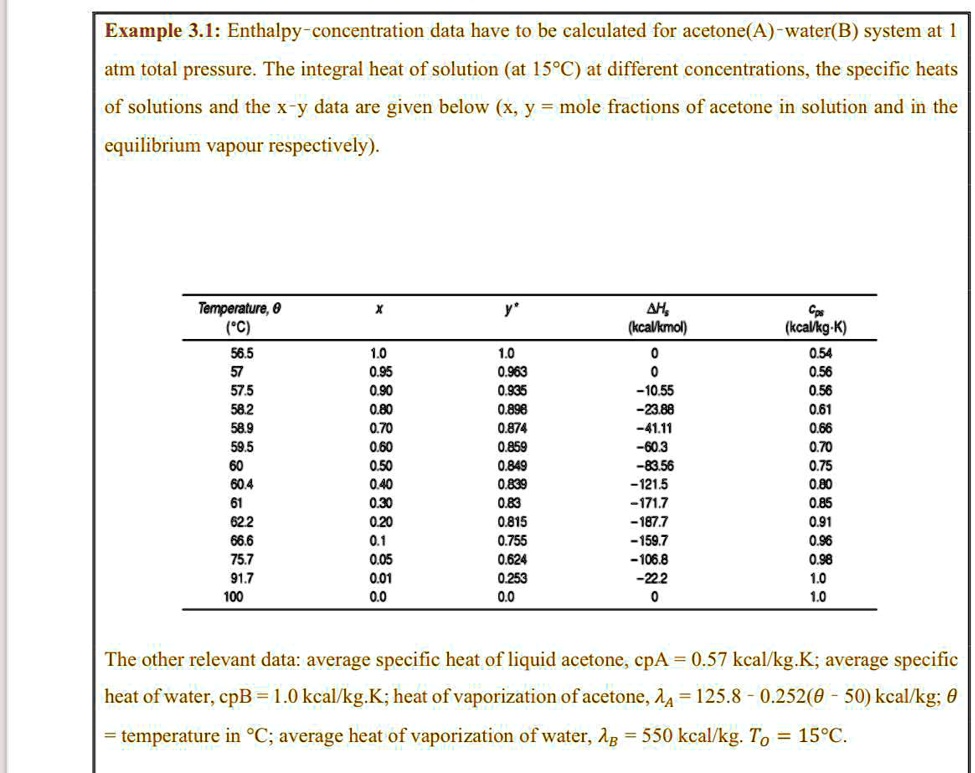
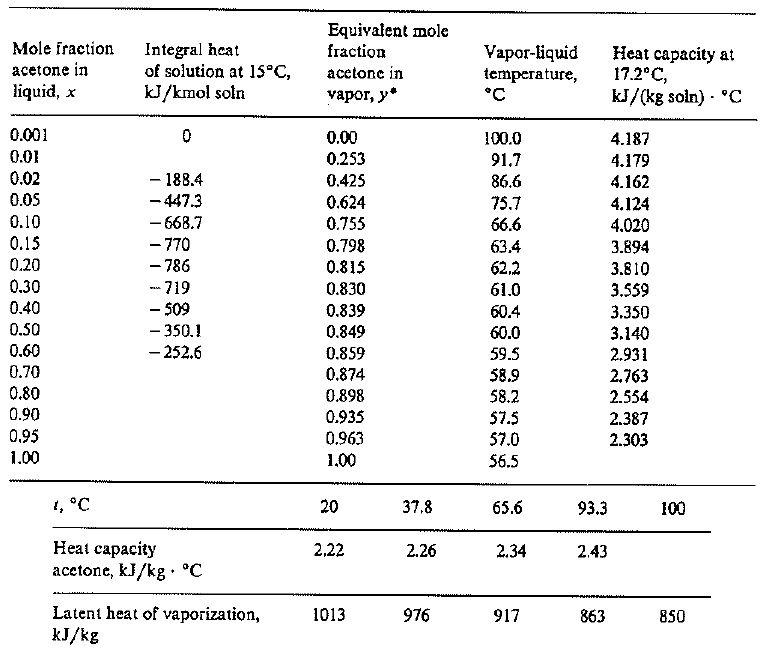
SOLVED: Example 3.1: Enthalpy-concentration data have to be calculated for acetone-water system at 1 atm total pressure. The integral heat of solution at 15°C at different concentrations, the specific heats of solutions,

SOLVED: I need the answer by using (Aspen HYSYS). Calculate the vapor pressure and latent heat of vaporization of acetone at 450 K.