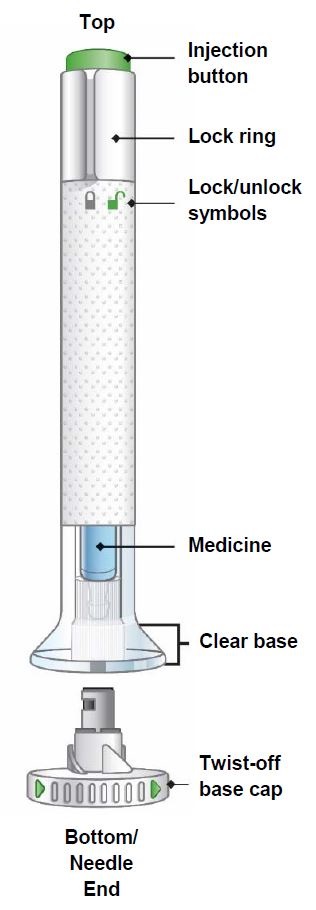
Instructions for use Taltz® 80 mg solution for injection in pre-filled pen ixekizumab Before using your pre-filled pen: Import
Lillys Taltz® (v) Receives U.S. FDA Approval for the Treatment of Pediatric Patients with Moderate to Severe Plaque Psoriasis
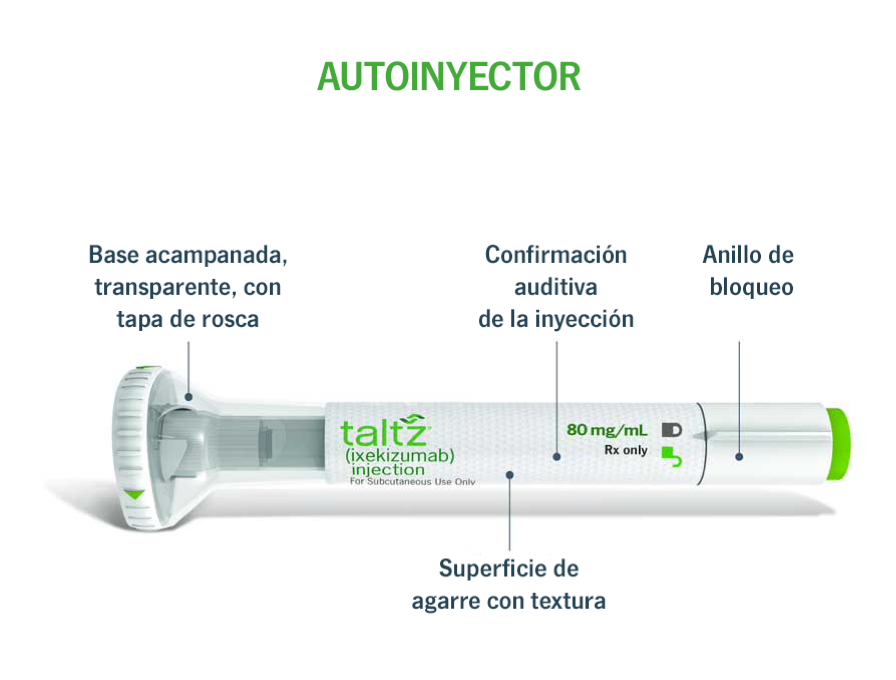
INSTRUCTIONS FOR USE How to use the TALTZ prefilled autoinjector PrTALTZ® (ixekizumab) injection 80 mg/mL www.lilly.ca PLEASE R
Instructions for use Taltz® 80 mg solution for injection in pre-filled pen ixekizumab Before using your pre-filled pen: Import
INSTRUCTIONS FOR USE How to use the TALTZ prefilled autoinjector PrTALTZ® (ixekizumab) injection 80 mg/mL www.lilly.ca PLEASE R
INSTRUCTIONS FOR USE How to use the TALTZ prefilled autoinjector PrTALTZ® (ixekizumab) injection 80 mg/mL www.lilly.ca PLEASE R
Instructions for use Taltz® 80 mg solution for injection in pre-filled pen ixekizumab Before using your pre-filled pen: Import