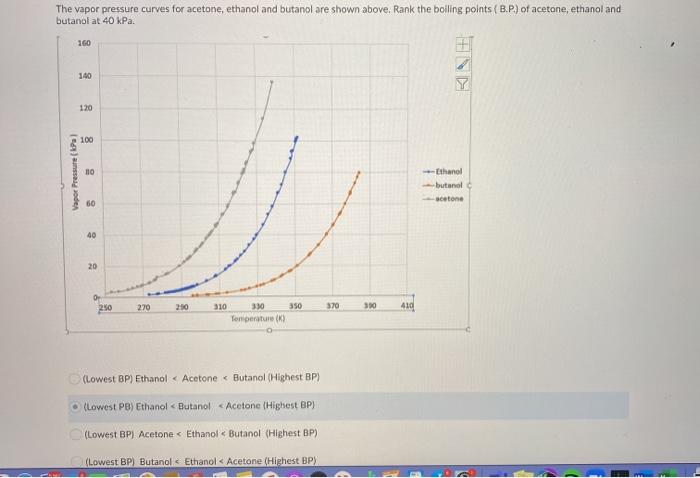
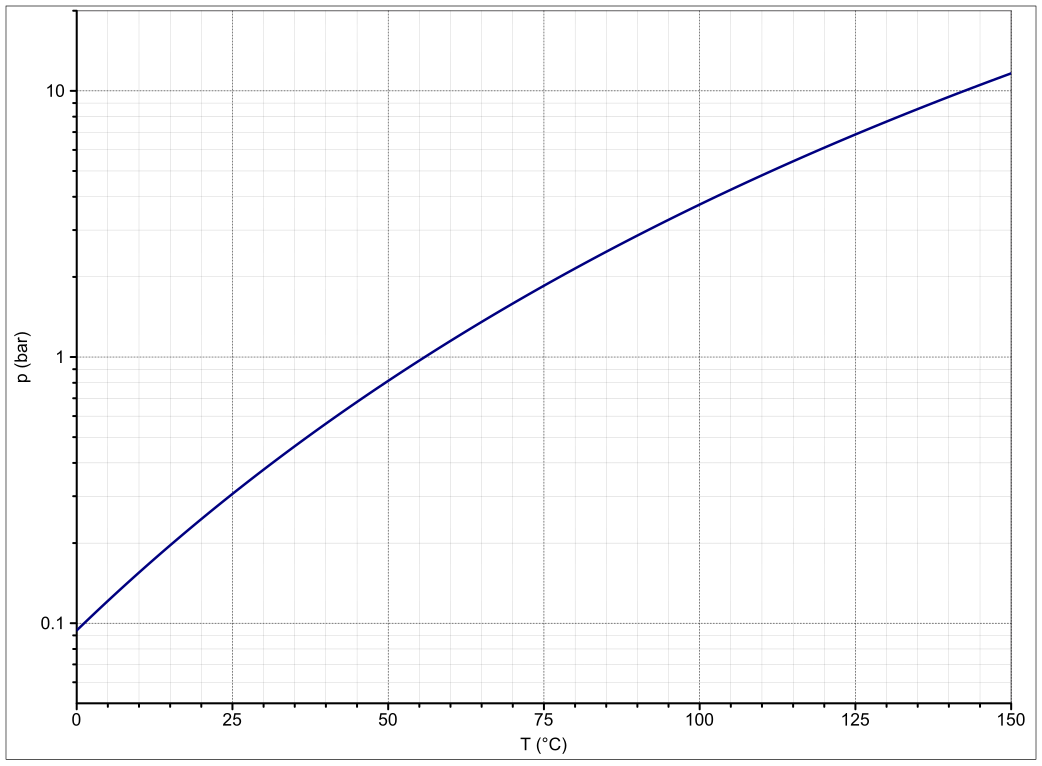
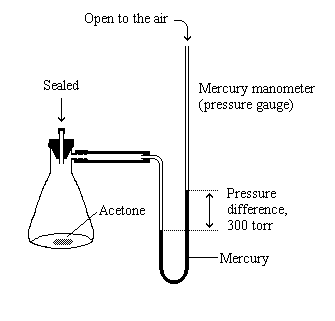
SOLVED: The vapor pressure of pure acetone (propanone) is 266 torr. When a non-volatile solute is added, the vapor pressure of acetone above the solution falls to 212 torr. What is the

The vapor pressure of acetone at `20^(@)C` is 185 torr. When `1.2 g` of a non-volatile solute - YouTube

Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot
The vapour pressure of acetone at 20°C is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at 20°C, - Sarthaks eConnect | Largest Online Education Community

The vapour pressure of acetone at `20^(@)C` is 185 torr. When `1.2 g` of non-volatile substance - YouTube
How to calculate the boiling point of a mixture made up of 2 liquids ( acetone+water in the same ratios) - Quora
The vapour pressure of acetone at 20C is 185 torr. When 1.2 g of a non-voiatile substance was dissolved in 100 g of acetone at 20C, its vapour pressure was 183 torr.

Vapour-liquid equilibrium of acetone-CO2 mixtures of different compositions at the vicinity of the critical point - ScienceDirect
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

The saturation vapor pressure of the benzene/acetone mixture (C 6 H 6 +... | Download Scientific Diagram